Module 2: Electrolysis (The Electrolytic Cell)
In Module 1, we learned that redox reactions involve the transfer of electrons. In a galvanic (voltaic) cell, this transfer happens spontaneously and generates electrical energy. In an electrolytic cell, we do the opposite: we supply electrical energy to drive a non-spontaneous redox reaction. This process is called electrolysis.
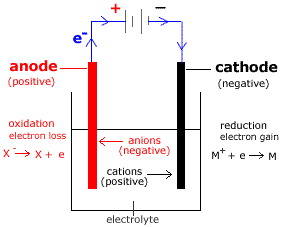
1. Components of an Electrolytic Cell
Every electrolytic cell consists of four essential parts:
Power Supply (Battery)
Provides the direct current (DC) that forces electrons to move.
Note: Positive terminal → anode; Negative terminal → cathode.
Electrolyte
An ionic compound (molten or in solution) that conducts electricity.
Ions move toward the electrodes during electrolysis.
Electrodes
Conducting rods (often metal or graphite) that allow electrons to enter or leave the electrolyte.
Electrolytic Tank
An inert vessel that holds the electrolyte and electrodes.
Usually open or designed to allow collection of gases.
1.1 Electrolytes: Strong vs. Weak
Strong Electrolyte
Dissociates completely into ions in solution.
Examples: \( NaCl \), \( H_2SO_4 \), \( KOH \)
Effect: Allows a large current to flow.
Weak Electrolyte
Dissociates only partially in solution.
Examples: Acetic acid, ammonia solution
Effect: Conducts electricity poorly.
Important: In electrolysis, strong electrolytes are usually used to ensure sufficient ion movement.
1.2 Electrodes: Inert vs. Active
Inert Electrodes
Examples: Platinum (\(Pt\)), graphite (carbon)
Behavior: Do not participate chemically; they only provide a surface for electron transfer.
Active Electrodes
Examples: Copper, silver, nickel
Behavior: Can be oxidized or reduced during electrolysis.
Commonly used in electroplating and refining.
The choice of electrode material significantly affects which reactions occur at the surface.